 Therefore, there are various non-equivalent definitions of atomic radius. However, this assumes the atom to exhibit a spherical shape, which is only obeyed for atoms in vacuum or free space. The atomic radius of a chemical element is a measure of the distance out to which the electron cloud extends from the nucleus. Transcribed image text: Question 13 (4 points) Arrange the following atoms in order of increasing atomic radius: N, K, As, Fr As the attractive force on the electrons in the outermost shell decreases. The number of shells increases hence screening effect increases. Explanation: As we move down the group atomic number increases, hence the nuclear charges increases. It must be noted, atoms lack a well-defined outer boundary. Trend: The atomic size increases down the group. The atomic radius of Francium atom is 260pm (covalent radius). 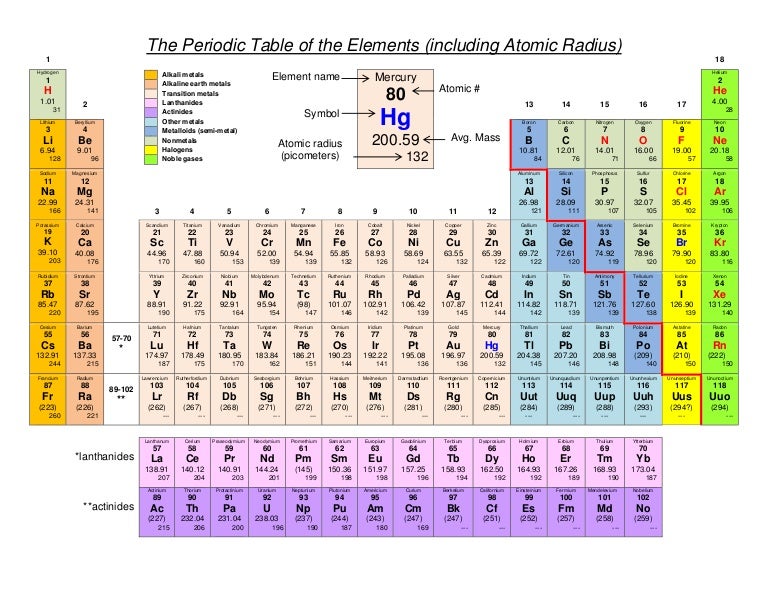 It must be noted, atoms lack a well-defined outer boundary. Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally-occuring isotopes and their abundance. Atomic radius or Atomic Radii is the total distance from the nucleus of an atom to the outermost orbital of its electron. The atomic radius of Francium atom is 260pm (covalent radius). The atomic mass is carried by the atomic nucleus, which occupies only about 10 -12 of the total volume of the atom or less, but it contains all the positive charge and at least 99.95% of the total mass of the atom. 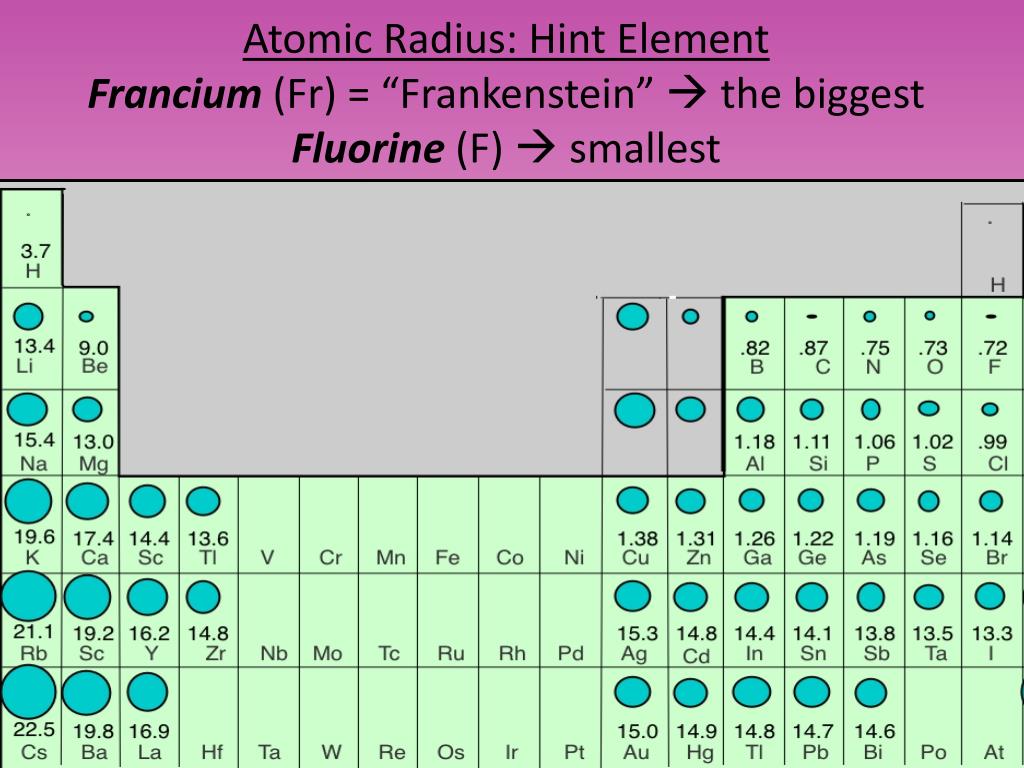 The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. Mass numbers of typical isotopes of Francium are 223. On separate sheets of graph paper plot Group 1 atomic symbol (Li Fr x-axis) vs. Up to date, curated data provided by Mathematica s ElementData function from Wolfram Research, Inc. Isotopes are nuclides that have the same atomic number and are therefore the same element, but differ in the number of neutrons. The difference between the neutron number and the atomic number is known as the neutron excess: D = N – Z = A – 2Z.įor stable elements, there is usually a variety of stable isotopes. Neutron number plus atomic number equals atomic mass number: N+Z=A. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol N. 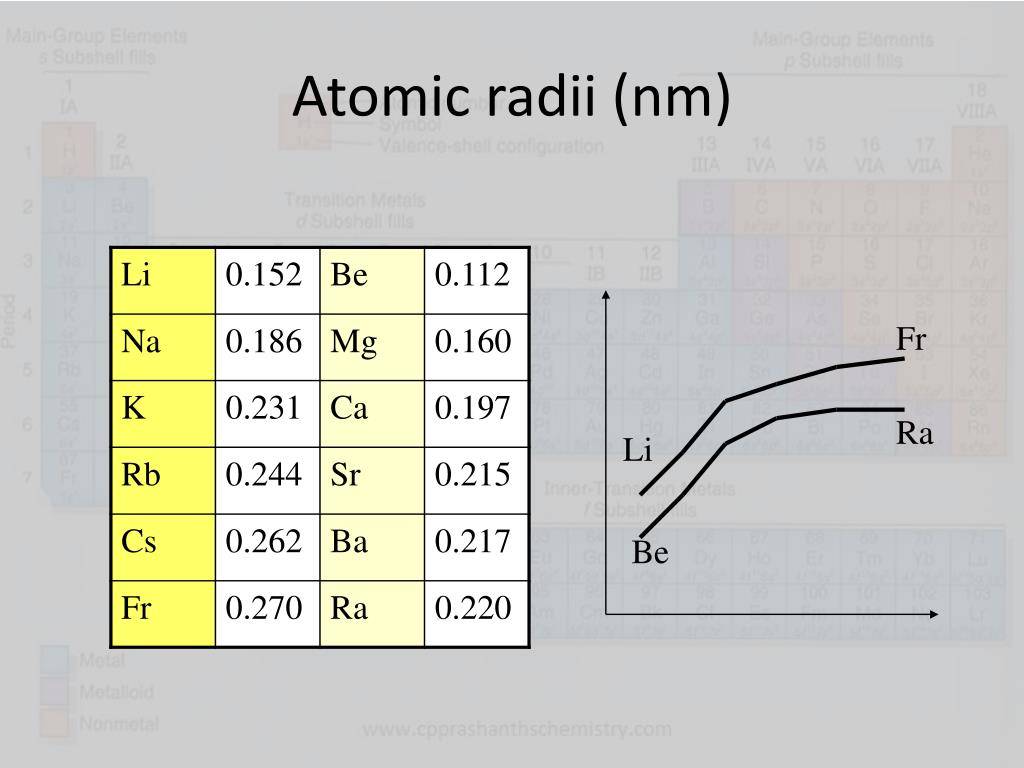 The total electrical charge of the nucleus is therefore +Ze, where e (elementary charge) equals to 1,602 x 10 -19 coulombs. Total number of protons in the nucleus is called the atomic number of the atom and is given the symbol Z. Interest in francium comes from the theoretical indications tha its heavy, unstable nucleus would provide a sensitive test for electron-quark interactions ( weak interaction).Atomic Number – Protons, Electrons and Neutrons in Franciumįrancium is a chemical element with atomic number 87 which means there are 87 protons in its nucleus. When elements are arranged in order of increasing atomic size. Further work should yield details about its atomic structure. Because all known isotopes of francium are highly unstable, knowledge of the chemical properties of this element comes from radiochemical techniques. 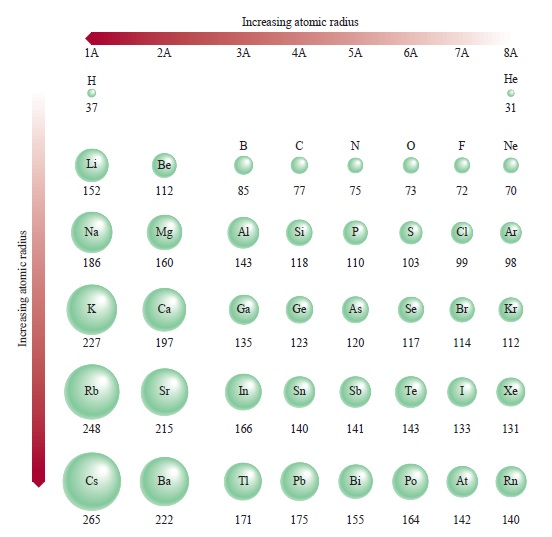 This is the only isotope of francium occurring in nature. The fluorescence from a collection of about 10,000 such atoms was bright enough to be recorded by a video camera. The longest lived 223 Fr (Ac, K), a daughter of 227 Ac, has a half-life of 22 min. About 1000 of these atoms at a time were trapped in a glass chamber by the combined action of six lasers at 718 nm and a magnetic field. In early 1996 it was manufactured by bombarding hot gold targets with oxygen-18 atoms, producing Fr-210. In nature it is found only in trace amounts in uranium deposits where it is produced in the uranium-235 decay series. It emits a beta particle of 1.1 MeV energy.įrancium is the least stable of the first 103 elements, making the measurement of its properties a considerable challenge. Compare and contrast ionization energy and atomic radius. This is due to the extra screening by the 3d electrons which. a) In group 1, which metal is the most active b) Metallic activity tends to (increase. All its isotopes are radioactive and short-lived its longest-lived isotope, francium-223, has a half-life of 22 minutes. Fe, Co and Ni are transition metals in the same period where the atomic radii do not vary much. Francium is the heaviest of the alkali metals and closely resembles cesium in its chemical properties. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
